 Relevant CMS manual instructions and policies may be found in the following Internet-Only Manuals (IOMs) published on the CMS Web site: Please review and understand them and apply the medical necessity provisions in the policy within the context of the manual rules. All providers who report services for Medicare payment must fully understand and follow all existing laws, regulations and rules for Medicare payment for Spinal Cord Stimulation (Dorsal Column Stimulation) and must properly submit only valid claims for them. Neither Medicare payment policy rules nor this LCD replace, modify or supersede applicable state statutes regarding medical practice or other health practice professions acts, definitions and/or scopes of practice. Federal statute and subsequent Medicare regulations regarding provision and payment for medical services are lengthy. This LCD supplements but does not replace, modify or supersede existing Medicare applicable National Coverage Determinations (NCDs) or payment policy rules and regulations for Spinal Cord Stimulation (Dorsal Column Stimulation). Not endorsed by the AHA or any of its affiliates. 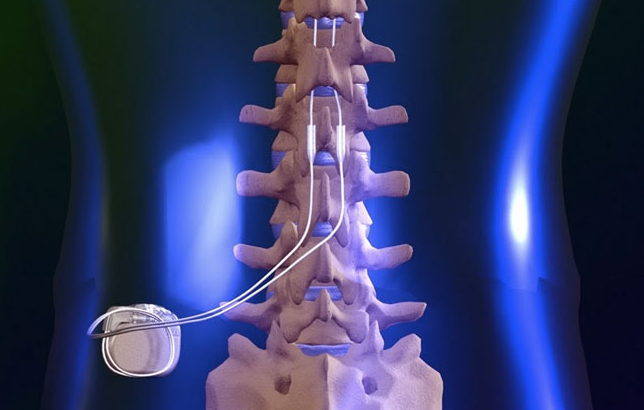
Presented in the material do not necessarily represent the views of the AHA. Preparation of this material, or the analysis of information provided in the material. The American Hospital Association (the "AHA") has not reviewed, and is not responsible for, the completeness orĪccuracy of any information contained in this material, nor was the AHA or any of its affiliates, involved in the 
Resale and/or to be used in any product or publication creating any modified or derivative work of the UB‐04 Manual and/or codes and descriptions Īnd/or making any commercial use of UB‐04 Manual or any portion thereof, including the codes and/or descriptions, is onlyĪuthorized with an express license from the American Hospital Association. Making copies or utilizing the content of the UB‐04 Manual, including the codes and/or descriptions, for internal purposes, If an entity wishes to utilize any AHA materials, please contact the AHA at 312‐893‐6816. AHA copyrighted materials including the UB‐04 codes andĭescriptions may not be removed, copied, or utilized within any software, product, service, solution or derivative work 
No portion of the American Hospital Association (AHA) copyrighted materials contained within this publication may beĬopied without the express written consent of the AHA. All rights reserved.Ĭopyright © 2023, the American Hospital Association, Chicago, Illinois. The AMA assumes no liability for data contained or not contained herein.Ĭurrent Dental Terminology © 2023 American Dental Association. The AMA does not directly or indirectly practice medicine or dispense medical services. Applicable FARS/HHSARS apply.įee schedules, relative value units, conversion factors and/or related components are not assigned by the AMA, are not part of CPT, and the AMA is not The procedure is usually safe, but may be associated with certain risks including change in electrode position, breakage, battery failure, bleeding, infection and development of tolerance.ĭorsal column stimulators do not treat the underlying cause of pain, but with its use, the intensity, frequency and duration of pain are usually reduced, lowering the need for pain medication and sometimes surgery.AMA CPT / ADA CDT / AHA NUBC Copyright StatementĬPT codes, descriptions and other data only are copyright 2023 American Medical Association. What are the Associated Risks and Complications of Dorsal Column Stimulator? Electrical impulses are generated at the pulse generator by remote control and are transmitted to the electrodes. The remaining components are connected externally. This is done either surgically or through the skin under radiographic guidance. Depending on the location of your pain, the electrodes are placed in the neck, upper or lower back region of the spinal cord. How is the Procedure of Dorsal Column Stimulator Performed?Ī dorsal column stimulator consists of electrodes, a battery-operated pulse generator, a remote control device and connecting wires. DCS acts either by blocking the pain impulse pathway or by stimulating the production of endorphins (chemical substances produced by the body that decrease the sensation of pain). It is used to treat chronic disabling pain such as failed back surgery syndrome and complex regional pain syndrome, which show little or no improvement with other treatments such as medication or surgery. The dorsal column stimulator (DCS) or spinal column stimulator is a device that relieves chronic pain associated with spinal nerves through electrical stimulation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
